Boron
2008/9 Schools Wikipedia Selection. Related subjects: Chemical elements
|
|||||||||||||||||||||||||
| General | |||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
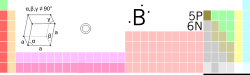
| Name, symbol, number | boron, B, 5 | ||||||||||||||||||||||||
| Chemical series | metalloids | ||||||||||||||||||||||||
| Group, period, block | 13, 2, p | ||||||||||||||||||||||||
| Appearance | black/brown |
||||||||||||||||||||||||
| Standard atomic weight | 10.811 (7) g·mol−1 | ||||||||||||||||||||||||
| Electron configuration | 1s2 2s2 2p1 | ||||||||||||||||||||||||
| Electrons per shell | 2, 3 | ||||||||||||||||||||||||
| Physical properties | |||||||||||||||||||||||||
| Phase | solid | ||||||||||||||||||||||||
| Density (near r.t.) | 2.34 g·cm−3 | ||||||||||||||||||||||||
| Liquid density at m.p. | 2.08 g·cm−3 | ||||||||||||||||||||||||
| Melting point | 2349 K (2076 ° C, 3769 ° F) |
||||||||||||||||||||||||
| Boiling point | 4200 K (3927 ° C, 7101 ° F) |
||||||||||||||||||||||||
| Heat of fusion | 50.2 kJ·mol−1 | ||||||||||||||||||||||||
| Heat of vaporization | 480 kJ·mol−1 | ||||||||||||||||||||||||
| Specific heat capacity | (25 °C) 11.087 J·mol−1·K−1 | ||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| Atomic properties | |||||||||||||||||||||||||
| Crystal structure | rhombohedral | ||||||||||||||||||||||||
| Oxidation states | 4, 3, 1 (mildly acidic oxide) |
||||||||||||||||||||||||
| Electronegativity | 2.04 (Pauling scale) | ||||||||||||||||||||||||
| Ionization energies ( more) |
1st: 800.6 kJ·mol−1 | ||||||||||||||||||||||||
| 2nd: 2427.1 kJ·mol−1 | |||||||||||||||||||||||||
| 3rd: 3659.7 kJ·mol−1 | |||||||||||||||||||||||||
| Atomic radius | 85 pm | ||||||||||||||||||||||||
| Atomic radius (calc.) | 87 pm | ||||||||||||||||||||||||
| Covalent radius | 82 pm | ||||||||||||||||||||||||
| Miscellaneous | |||||||||||||||||||||||||
| Magnetic ordering | nonmagnetic | ||||||||||||||||||||||||
| Electrical resistivity | (20 °C) 1.5×104Ω·m | ||||||||||||||||||||||||
| Thermal conductivity | (300 K) 27.4 W·m−1·K−1 | ||||||||||||||||||||||||
| Thermal expansion | (25 °C) 5–7 µm·m−1·K−1 | ||||||||||||||||||||||||
| Speed of sound (thin rod) | (20 °C) 16200 m/s | ||||||||||||||||||||||||
| Bulk modulus | (β form) 185 GPa | ||||||||||||||||||||||||
| Mohs hardness | 9.3 | ||||||||||||||||||||||||
| Vickers hardness | 49000 MPa | ||||||||||||||||||||||||
| CAS registry number | 7440-42-8 | ||||||||||||||||||||||||
| Selected isotopes | |||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| References | |||||||||||||||||||||||||
Boron (pronounced /ˈbɔərɒn/) is a chemical element with atomic number 5 and the chemical symbol B. Boron is a trivalent nonmetallic element which occurs abundantly in the evaporite ores borax and ulexite. Boron is never found as a free element in nature.
Several allotropes of boron exist; amorphous boron is a brown powder, though crystalline boron is black, hard (9.3 on Mohs' scale), and a weak conductor at room temperature. Elemental boron is used as a dopant in the semiconductor industry, while boron compounds play important roles as light structural materials, nontoxic insecticides and preservatives, and reagents for chemical synthesis.
Boron is an essential plant nutrient, although higher soil concentrations of boron may also be toxic to plants. As an ultratrace element, boron is necessary for the optimal health of rats and presumably other mammals, though its physiological role in animals is poorly understood.
Characteristics
Brown amorphous boron is a product of certain chemical reactions. It contains boron atoms randomly bonded to each other without long range order.
Crystalline boron, a very hard black material with a high melting point, exists in many polymorphs. Two rhombohedral forms, α-boron and β-boron containing 12 and 106.7 atoms in the rhombohedral unit cell respectively, and 50-atom tetragonal boron are the three most characterised crystalline forms.
Optical characteristics of crystalline/elemental boron include the transmittance of infrared light. At standard temperatures, elemental boron is a poor electrical conductor, but is a good electrical conductor at high temperatures.
Chemically boron is electron-deficient, possessing a vacant p-orbital. It is an electrophile. Compounds of boron often behave as Lewis acids, readily bonding with electron-rich substances to compensate for boron's electron deficiency. The reactions of boron are dominated by such requirement for electrons. Also, boron is the least electronegative non-metal, meaning that it is usually oxidized (loses electrons) in reactions.
Boron is also similar to carbon with its capability to form stable covalently bonded molecular networks. Boron is also used for heat resistant alloys. Boron forms a polyatomic B(II), such as B2F4.
Applications
- In automobiles: it is proposed that by reacting water with elemental boron, hydrogen could be produced to be burnt in an internal combustion engine or fed to a fuel cell to generate electricity.
10B and 11B NMR spectroscopy
both 10B (18.8 percent) and 11B (81.2 percent) possess nuclear spin; that of boron-10 has a value of 3 and that of boron-11, 3/2. These isotopes are, therefore, of use in nuclear magnetic resonance spectroscopy; and spectrometers specially adapted to detecting the boron-11 nucleus are available commercially. The boron-10 and boron-11 nuclei also cause splitting in the resonances of attached nuclei.
10B enriched boron
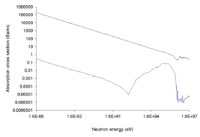
The 10B isotope is good at capturing thermal neutrons. Natural boron is about 20% 10B and 80%11B. The nuclear industry enriches natural boron to nearly pure 10B. The waste product, or depleted boron, is nearly pure 11B. 11B is a candidate as a fuel for aneutronic fusion and is used in the semiconductor industry. Enriched boron or 10B is used in both radiation shielding and in boron neutron capture therapy. In the later, a compound containing 10B is attached to a muscle near a tumor. The patient is then treated with a relatively low dose of thermal neutrons. This causes energetic and short range alpha radiation from the boron to bombard the tumor.
In nuclear reactors, 10B is used for reactivity control and in emergency shutdown systems. It can serve either function in the form of borosilicate control rods or as boric acid. In pressurized water reactors, boric acid is added to the reactor coolant when the plant is shut down for refueling. It is then slowly filtered out over many months as fissile material is used up and the fuel becomes less reactive.
In future manned interplanetary spacecraft, 10B has a theoretical role as structural material (as boron fibers or BN nanotube material) which also would serve a special role in the radiation shield. One of the difficulties in dealing with cosmic rays which are mostly high energy protons, is that some secondary radiation from interaction of cosmic rays and spacecraft structural materials, is in the form of high energy spallation neutrons. Such neutrons can be moderated by materials high in light elements such as structural polyethylene, but the moderated neutrons continue to be a radiation hazard unless actively absorbed in a way which dumps the absorption energy in the shielding, far away from biological systems. Among light elements that absorb thermal neutrons, 6Li and 10B appear as potential spacecraft structural materials able to do double duty in this regard.
11B depleted boron
Cosmic radiation produces secondary neutrons when it hits spacecraft structures. Neutrons produce fission in 10B if it is present in the spacecraft's semiconductors. This produces a gamma ray, an alpha particle, and a lithium ion. The resultant fission products may then dump charge into nearby chip structures, causing data loss (bit flipping, or single event upset). In radiation hardened semiconductor designs, one measure is to use depleted boron which is greatly enriched in 11B and contains almost no 10B. 11B is largely immune to radiation damage. Depleted boron is a by-product of the nuclear industry.
11B is also a candidate as a fuel for aneutronic fusion. When struck by a proton of about 500 Kev, it produces three alpha particles and 8.7 Mev of energy. Most other fusion reactions involving hydrogen and helium produce penetrating neutron radiation. This induces long term radioactivity in reactor structures and weakens them, as well as endangering operating personel. The alpha particles from 11B fusion can be turned directly into electric power and all radiation stops as soon as the reactor is turned off.
Market trend
Estimated global consumption of boron rose to a record 1.8 million tonnes of B2O3 in 2005 following a period of strong growth in demand from Asia, Europe and North America. Boron mining and refining capacities are considered to be adequate to meet expected levels of growth through the next decade. The form in which boron is consumed has changed in recent years. The use of beneficiated ores like colemanite has declined following concerns over arsenic content. Consumers have moved towards the use of refined borates or boric acid that have a lower pollutant content. The average cost of crystalline boron is $5/g.
Increasing demand for boric acid has led a number of producers to invest in additional capacity. Eti Mine opened a new 100,000 tonnes per year capacity boric acid plant at Emet in 2003. Rio Tinto increased the capacity of its Boron plant from 260,000 tonnes per year in 2003 to 310,000 tonnes per year by May 2005, with plans to grow this to 366,000 tonnes per year in 2006.
Chinese boron producers have been unable to meet rapidly growing demand for high quality borates. This has led to imports of disodium tetraborate growing by a hundredfold between 2000 and 2005 and boric acid imports increasing by 28% per year over the same period.
The rise in global demand has been driven by high rates of growth in fibreglass and borosilicate production. A rapid increase in the manufacture of reinforcement-grade fiberglass in Asia with a consequent increase in demand for borates has offset the development of boron-free reinforcement-grade fiberglass in Europe and the USA. The recent rises in energy prices can be expected to lead to greater use of insulation-grade fibreglass, with consequent growth in the use of boron.
Roskill Consulting Group forecasts that world demand for boron will grow by 3.4% per year to reach 21 million tonnes by 2010. The highest growth in demand is expected to be in Asia where demand could rise by an average 5.7% per year.
Boron compounds
The most economically important compounds of boron
- Sodium tetraborate pentahydrate (Na2B4O7 · 5H2O), which is used in large amounts in making insulating fibreglass and sodium perborate bleach,
- Orthoboric acid (H3BO3) or boric acid, used in the production of textile fibreglass and flat panel displays or eye drops, among many uses, and
- Sodium tetraborate decahydrate (Na2B4O7 · 10H2O) or borax, used in the production of adhesives, in anti-corrosion systems and many other uses.
- Boron nitride is a material in which the extra electron of nitrogen (with respect to carbon) in some ways compensates for boron's deficiency of an electron.
- Boron reacts with ammonia at high temperatures to give a compound called borazole (B3N3H6), also known as inorganic benzene.
Of the several hundred uses of boron compounds, especially notable uses
- Boron is an essential plant micronutrient.
- Because of its distinctive green flame, amorphous boron is used in pyrotechnic flares.
- Boric acid is an important compound used in textile products.
- Boric acid is also traditionally used as an insecticide, notably against ants, fleas, and cockroaches.
- Borax is sometimes found in laundry detergent.
- Boron filaments are high-strength, lightweight materials that are chiefly used for advanced aerospace structures as a component of composite materials, as well as limited production consumer and sporting goods such as golf clubs and fishing rods.
- Boron is used as a melting point depressant in nickel-chromium braze alloys.
- Boron slurry is used as an energetic material with very high energy density like rocket fuels and jet engines.
- Boron compounds show promise in treating arthritis.
Hardest boron compound
The hardest Boron compound is created synthetically. Rhenium diboride (ReB2) can actually scratch diamond, giving it a higher than 10 rank in the Mohs scale of mineral hardness and making it one of the three hardest known substances - the other two being ultrahard fullerite and aggregated diamond nanorods.
History
Compounds of boron (Arabic Buraq from Persian Burah from Turkish Bor) have been known of for thousands of years. In early Egypt, mummification depended upon an ore known as natron, which contained borates as well as some other common salts. Borax glazes were used in China from 300 AD, and boron compounds were used in glassmaking in ancient Rome.
The element was not isolated until 1808 by Sir Humphry Davy, Joseph Louis Gay-Lussac, and Louis Jacques Thénard, to about 50 percent purity, by the reduction of boric acid with sodium or magnesium. These men did not recognize the substance as an element. It was Jöns Jakob Berzelius in 1824 who identified boron as an element. The first pure boron was produced by the American chemist W. Weintraub in 1909, although this is disputed by some researchers.
Occurrence
Turkey and the United States are the world's largest producers of boron. Turkey has almost 72% of the world’s boron potential and boron reserves. Boron does not appear in nature in elemental form but is found combined in borax, boric acid, colemanite, kernite, ulexite and borates. Boric acid is sometimes found in volcanic spring waters. Ulexite is a borate mineral that naturally has properties of fibre optics.
Economically important sources are from the ore rasorite (kernite) and tincal (borax ore) which are both found in the Mojave Desert of California, with borax being the most important source there. The largest borax deposits are found in Central and Western Turkey including the provinces of Eskişehir, Kütahya and Balıkesir.
Commercial production of the free element
Pure elemental boron is not easy to prepare. The earliest methods used involve reduction of boric oxide with metals such as magnesium or aluminium. However the product is almost always contaminated with metal borides. (The reaction is quite spectacular though.) Pure boron can be prepared by reducing volatile boron halogenides with hydrogen at high temperatures. The highly pure boron, for the use in semiconductor industry, is produced by the decomposition of diborane at high temperatures and then further purified with the Czochralski process.
Boron in biology
A boron-containing natural antibiotic, boromycin, isolated from streptomyces, is known.
Boron is an essential plant nutrient, required primarily for maintaining the integrity of cell walls. Conversely, high soil concentrations of > 1.0 ppm can cause marginal and tip necrosis in leaves as well as poor overall growth performance. Levels as low as 0.8 ppm can cause these same symptoms to appear in plants particularly sensitive to boron in the soil. Nearly all plants, even those somewhat tolerant of boron in the soil, will show at least some symptoms of boron toxicity when boron in the soil is greater than 1.8 ppm. When boron in the soil exceeds 2.0 ppm, few plants will perform well. Plants sensitive to boron in the soil may not survive. When boron levels in plant tissue exceed 200 ppm symptoms of boron toxicity are likely to appear.
As an ultratrace element, boron is necessary for the optimal health of rats, although it is necessary in such small amounts that ultrapurified foods and dust filtration of air is necessary to show the effects of boron deficiency, which manefest as poor coat/hair quality. Presumably, boron is necessary to other mammals. No deficiency syndrome in humans has been described. Small amounts of boron occur widely in the diet, and the amounts needed in the diet would, by analogy with rodent studies, be very small. The exact physiological role of boron in the animal kingdom is poorly understood.
Boron occurs in all foods produced from plants. Since 1989 its nutritional value has been argued. It is thought that boron plays several biochemical roles in animals, including humans. The U.S. Department of agriculture conducted an experiment in which postmenopausal women took 3 mg of boron a day. The results showed that supplemental boron reduced excretion of calcium by 44%, and activated estrogen and vitamin D. However, whether these effects were conventionally nutritional, or medicinal, could not be determined.
The US National Institute of Health quotes this source:
- Total daily boron intake in normal human diets ranges from 2.1–4.3 mg boron/kg body weight (bw)/day. "Total boron". Zook EG and Lehman J. J. Assoc. Off Agric. Chem. 48: 850-5 (1965).
Analytical quantification
For determination of boron content in food or materials the colorimetric curcumin method is used. Boron has to be transferred to boric acid or borates and on reaction with curcumin in acidic solution a red colored boron- chelate complex, rosocyanine, is formed.
Isotopes
Boron has two naturally-occurring and stable isotopes, 11B (80.1%) and 10B (19.9%). The mass difference results in a wide range of δ11B values in natural waters, ranging from -16 to +59. There are 13 known isotopes of boron, the shortest-lived isotope is 7B which decays through proton emission and alpha decay. It has a half-life of 3.26500x10-22 s. Isotopic fractionation of boron is controlled by the exchange reactions of the boron species B(OH)3 and B(OH)4. Boron isotopes are also fractionated during mineral crystallization, during H2O phase changes in hydrothermal systems, and during hydrothermal alteration of rock. The latter effect species preferential removal of the 10B(OH)4 ion onto clays results in solutions enriched in 11B(OH)3 may be responsible for the large 11B enrichment in seawater relative to both oceanic crust and continental crust; this difference may act as an isotopic signature.
The exotic 17B exhibits a Nuclear halo.
Precautions
Elemental boron is nontoxic and common boron compounds such as borates and boric acid have low toxicity (approximately similar to table salt with the lethal dose being 2 to 3 grams per kg) and therefore do not require special precautions while handling. Some of the more exotic boron hydrogen compounds, however, are toxic as well as highly flammable and do require special handling care.