Nitrate
2008/9 Schools Wikipedia Selection. Related subjects: Chemical compounds
In inorganic chemistry, a nitrate is a salt of nitric acid with an ion composed of one nitrogen and three oxygen atoms (NO3−). In organic chemistry the esters of nitric acid and various alcohols are called nitrates.
Chemical properties
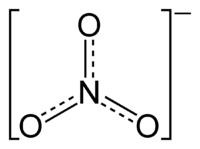
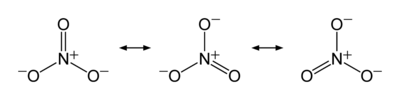
The nitrate ion is a polyatomic ion with the empirical formula NO3− and a molecular mass of 62.0049. It is the conjugate base of nitric acid, consisting of one central nitrogen atom surrounded by three identical oxygen atoms in a trigonal planar arrangement. The nitrate ion carries a formal charge of negative one, where each oxygen carries a −2/3 charge while the nitrogen carries a +1 charge, and is commonly used as an example of resonance. The three canonical structures of the nitrate ion are shown resonating below:
Almost all inorganic nitrate salts are soluble in water at standard temperature and pressure.
In organic chemistry a nitrate is a functional group with general chemical formula RONO2 where R stands for any organic residue. They are the esters of nitric acid and alcohols formed by nitroxylation. Examples are methyl nitrate formed by reaction of methanol and nitric acid, the nitrate of tartaric acid, and the inappropriately named nitroglycerin.
Related materials
Nitrates should not be confused with nitrites (NO2−) the salts of nitrous acid. Organic compounds containing the nitro functional group (which has the same formula and structure as the nitrate ion save that one of the O− atoms is replaced by the R group) are known as nitro compounds.
Effects on aquatic life
In freshwater or estuarine systems close to land, nitrate can reach high levels that can potentially cause the death of fish. While nitrate is much less toxic than ammonia or nitrite, levels over 30 ppm of nitrate can inhibit growth, impair the immune system and cause stress in some aquatic species. However, in light of inherent problems with past protocols on acute nitrate toxicity experiments, the extent of nitrate toxicity has been the subject of recent debate.
In most cases of excess nitrate concentrations, the principle pathway of entering aquatic systems is through surface runoff from agricultural or landscaped areas which have received excess nitrate fertilizer. These levels of nitrate can also lead to algae blooms, and when nutrients become limiting (such as potassium, phosphate or nitrate) then eutrophication can occur. As well as leading to water anoxia, these blooms may cause other changes to ecosystem function, favouring some groups of organisms over others. Consequently, as nitrates form a component of total dissolved solids, they are widely used as an indicator of water quality.
Nitrates are also a by-product of septic systems. Specifically, they are a naturally occurring chemical that is left after the break down or decomposition of animal or human waste. Water quality may also be affected through ground water resources that have a high number of septic systems in a watershed. Septics leach down into ground water resources or aquifers and supply near by bodies of water. Lakes that rely on ground water are often affected by nitrification through this process.